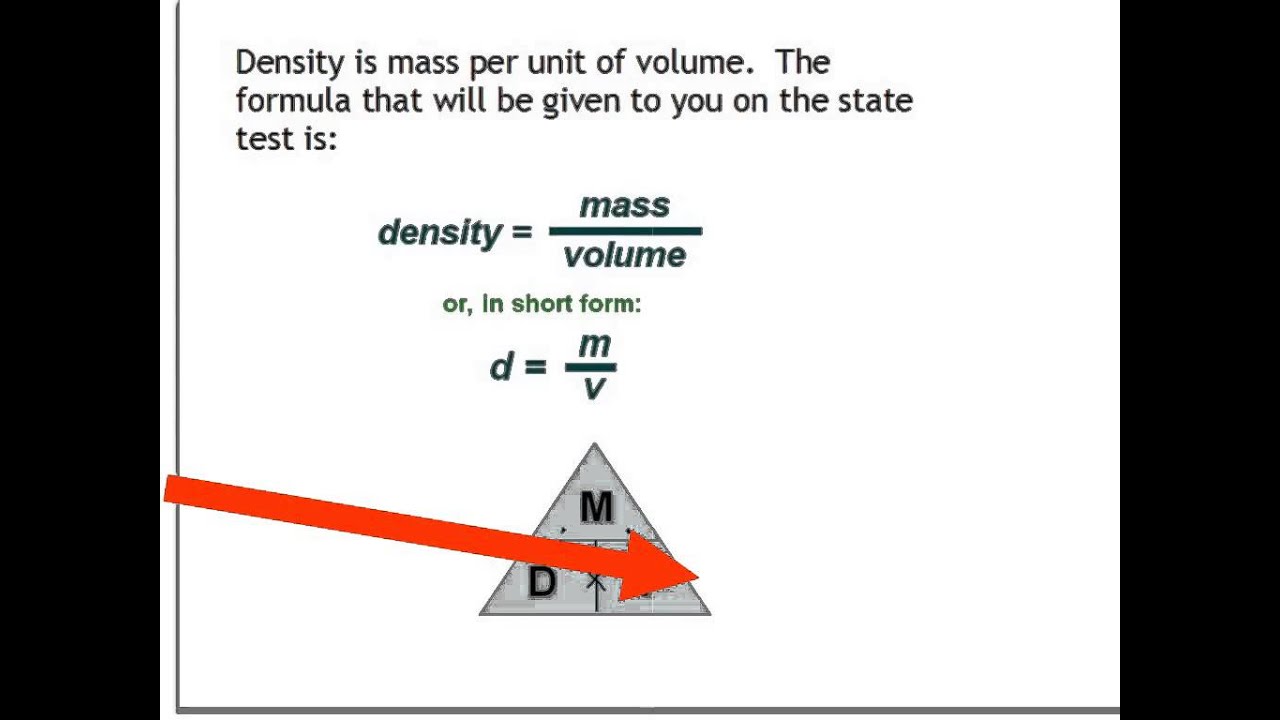
You'll have estimated the Earth's average density and come up with a result of 5,500 kg/m^3.Look up the Earth's volume in m^3, which is 1.1 X 10^21 m^3.Take note of the Earth's mass in kilogrammes, which is 6 X 10^24 kg.Because gases are substantially less dense than solids and liquids, their densities are frequently expressed in grammes per litre.ĥ. Density units can alternatively be expressed as g/mL because a cubic centimetre equals a millilitre. In millilitres, how do you write density?ĭensities of most solids and liquids are commonly given in grammes per cubic centimetre (g/cm^3). Saturn would float if it were placed in a body of water!Ĥ. This is a fraction of the density of water, which is 1,000 kg/m^3. Saturn has the lowest density among the eight planets in the Solar System, with 687 kg/m^3. That's d = m/v in equation form, where d is the density, m is the mass, and v is the object's volume. The tank calibrated for this calculation. The mass of an object divided by its volume is the formula for density. This app is used to find the Density at 15C (ASTM 53B) and also calculate the tank volume based on the cm reading. If two materials have the same volume, a material with a higher density will weigh more. There are exceptions, such as when the density of water increases between 0☌ and 4☌.Ī material's density is the amount of mass it possesses per unit volume. Temperature increases tend to reduce density since the volume will normally increase. When pressure rises, volume falls and density always rises. When it comes to gases, however, temperature and pressure have a significant impact on density. The density of solids and liquids normally changes very little. How do I calculate the density To calculate the density use the formula m/V, where m is the mass and V is the volume. It's important to note that pressure and temperature have an impact on density. It's a good idea to write down all of the numbers you're working with, including units, and then do a dimensional analysis to make sure the end output has the right units. There are many different ways to describe density, and if you don't use or convert it into the right units, you'll get the wrong answer. Therefore, it is critical to pay close attention to the units employed in density calculations. The formula for calculating density is simple. 
We have Archimedes to thank for that, who shouted the Greek phrase when he discovered the lies of the king's craftsman.The formula to calculate density is given as follows You might have heard the famous line, "Eureka!", which is associated with figuring something out. He found that the crown in question was, in fact, a fraud! It displaced more water than the pure gold one. He knew that silver had a lower density than gold and would, therefore, displace more water since the mass of the two crowns were the same. He placed the crown in question and a truly pure gold crown in a tub of water.  
He decided to test the crown in a similar manner. He recognized that his body mass displaced some of the water, and he then had the first thoughts about what a material's density could mean. When he got in the tub, he noticed that some of the water spilled out, which got him thinking. This calculator can be used to calculate expansion volume when initial volume and initial and final densities for the liquid are known. Calculate density of different material from its mass (weight) and volume in english, metric, imperial or local units simply enter mass or weight of an. The king had a suspicion that the crown he had gotten was actually a fraud, mostly made of silver instead of gold. Online Thermal Cubic Expansion Calculator - Densities. In fact, he discovered as a by-product for a certain task he was charged with.Īs the historical story goes, Archimedes was tasked with determining whether or not the King of Syracuse's craftsman was lying to him about the purity of the golden crown presented to the king. by Archimedes, a rather famous Greek mathematician of the time. Density was first discovered in 250 B.C.E.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
